Morphology and volcanology Kimberlites occur as carrot shaped, vertical intrusions termed 'pipe'. This classic carrot shape is formed due to a complex intrusive process of kimberlitic magma that inherit a large proportion of both CO2 and H2O in the system which produces a deep explosive boiling stage that causes a significant amount of vertical flaring (Bergman, 1987). Kimberlite classification is based on the recognition of differing rock facies. These differing facies are associated with a particular style of magmatic activity, namely crater, diatreme and hypabyssal rocks (Clement and Skinner 1985, and Clement, 1982).
The morphology of kimberlite pipes, and the classical carrot shape, is the result of explosive diatreme volcanism from very deep mantle derived sources. These volcanic explosions produce vertical columns of rock that rise from deep magma reservoirs. The morphology of kimberlite pipes is varied but generally includes a sheeted dyke complex of tabular, vertically dipping feeder dykes in the root of the pipe which extends down to the mantle. Within 1.5-2 km of the surface the highly pressured magma explodes upwards and expands to form a conical to cylindrical diatreme, which erupts to the surface. The surface expression is rarely preserved but is usually similar to a maar volcano. The diameter of a kimberlite pipe at the surface is typically a few hundred meters to a kilometer.
Petrology Both the location and origin of kimberlitic magmas are areas of contention. Their extreme enrichment and geochemistry has led to a large amount of speculation about their origin, with models placing their source within the sub-continental lithospheric mantle (SCLM) or even as deep as the transition zone. The mechanism of enrichment has also been the topic of interest with models including partial melting, assimilation of subducted sediment or derivation from a primary magma source.
Historically, kimberlites have been subdivided into two distinct varieties termed 'basaltic' and 'micaceous' based primarily on petrographic observations (Wagner, 1914). This was later revised by Smith (1983) who re-named these divisions Group I and Group II based on the isotopic affinities of these rocks using the Nd, Sr and Pb systems. Mitchell (1995) later proposed that these group I and II kimberlites display such distinct differences, that they may not be as closely related as once thought. He showed that Group II kimberlites actually show closer affinities to lamproites than they do to Group I kimberlites. Hence, he reclassified Group II kimberlites as orangeites to prevent confusion.
Group I kimberlites Group I kimberlites are of CO2-rich ultramafic potassic igneous rocks dominated by a primary mineral assemblage of forsteritic olivine, magnesian ilmenite, chromian pyrope, almandine-pyrope, chromian diopside (in some cases subcalcic), phlogopite, enstatite and of Ti-poor chromite. Group I kimberlites exhibit a distinctive inequigranular texture cause by macrocrystic (0.5-10 mm) to megacrystic (10-200 mm) phenocrysts of olivine, pyrope, chromian diopside, magnesian ilmenite and phlogopite in a fine to medium grained groundmass.
The groundmass mineralogy, which more closely resembles a true composition of the igneous rock, contains forsteritic olivine , pyrope garnet, Cr-diopside , magnesian ilmenite and spinel .
Group II kimberlites Group-II kimberlites (or orangeites) are ultrapotassic, peralkaline rocks rich in volatiles (dominantly H2O). The distinctive characteristic of orangeites is phlogopite macrocrysts and microphenocrysts, together with groundmass micas that vary in composition from phlogopite to "tetraferriphlogopite" (anomalously Fe-rich phlogopite). Resorbed olivine macrocrysts and euhedral primary crystals of groundmass olivine are common but not essential constituents.
Characteristic primary phases in the groundmass include: zoned pyroxenes (cores of diopside rimmed by Ti-aegirine); spinel-group minerals (magnesian chromite to titaniferous magnetite); Sr- and REE-rich perovskite; Sr-rich apatite ; REE-rich phosphates (monazite , daqingshanite); potassian barian hollandite group minerals; Nb-bearing rutile and Mn-bearing ilmenite .
Kimberlitic indicator minerals Kimberlites are peculiar igneous rocks because they contain a variety of mineral species with peculiar chemical compositions. These minerals such as potassic richterite, chromian diopside (a pyroxene ), chromium spinels, magnesian ilmenite, and garnets rich in pyrope plus chromium are generally absent from most other igneous rocks, making them particularly useful as indicators for kimberlites.
These indicator minerals are generally sought in stream sediments in modern alluvial material. Their presence, when found, may be indicative of the presence of a kimberlite within the erosional watershed which has produced the alluvium.
Economic importance Kimberlites are the most important source of primary diamonds. Many kimberlite pipes also produce rich alluvial or eluvial diamond placer deposits. However, only about 1 in 200 kimberlite pipes contain gem-quality diamonds.
The deposits occurring at Kimberley, South Africa were the first recognized and the source of the name. The Kimberley diamonds were originally found in weathered kimberlite which was colored yellow by limonite, and so was called yellow ground. Deeper workings encountered less altered rock, serpentinized kimberlite, which miners call blue ground.
The blue and yellow ground were both prolific producers of diamonds. After the yellow ground had been exhausted, miners in the late 1800's accidentally cut into the blue ground and found gem quality diamonds in quantity. The economic importance of the time is that with flood of diamonds being found, the miners were undercutting each other's price of the diamonds and eventually decreased the diamonds value down to cost in a short time.
How are diamonds formed?
Author: Ravi Kewalramani
Content by courtesy of : Gem Sutra
The word "Diamond" comes from the greek word "Adamas", which means indestructible. It is the only gem known to man that is made of a single element, Carbon, besides graphite. Diamond is completely made of Carbon atoms (Chemical Composition - 'C') crystallised in a cubic (isometric) arrangement.
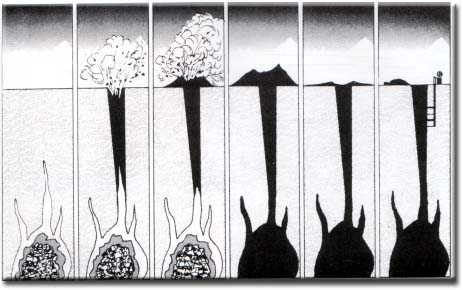
How and where are diamonds formed?
Diamonds form between 120-200 kms or 75-120 miles below the earth's surface. According to geologists the first delivery of diamonds was somewhere around 2.5 billion years ago and the most recent was 45 million years ago. That is a long time, my friend! According to science , the carbon that makes diamonds, comes from the melting of pre-existing rocks in the Earth's upper mantle. There is an abundance of carbon atoms in the mantle. Temperature changes in the upper mantle forces the carbon atoms to go deeper where it melts and finally becomes new rock, when the temperature reduces. If other conditions like pressure and chemistry is right then the carbon atoms in the melting crustal rock bond to build diamond crystals.
There is no guarantee that these carbon atoms will turn into diamonds. If the temperature rises or the pressure drops then the diamond crystals may melt partially or totally dissolve. Even if they do form, it takes thousands of years for those diamonds to come anywhere near the surface. Are you guys with me? A lot of information, huh! Let's continue!
Diamond's journey to the surface?
Diamond deposits are called Kimberlite Pipes or Blue Ground. These are also called Primary Mines. On the other hand, diamonds are also found at river beds. These are Alluvial Deposits.

So you see that Mother Nature has to toil for millions of years to make a diamond. When you own a piece of diamond, you own something which is a legend in the making. It has not been made in a factory just the other day. A diamond comes from the bosom of the earth. More interestingly not all the diamonds mined are made into jewelry. Only one fourth quantity that is mined is made into jewelry. Every 100 tons of mud produces one carat of a diamond. And might I add, this one carat is not one stone! It could be anything from 0.005 ct to 1 ct.
Would you like to see how a rough diamond looks? This image on your right is an example of a rough diamond. This particular one is called a 'Glassy'. Glassy is a rough diamond that comes out clean from the mines and does not require polishing. It is very rare to get a glassy. Diamonds come in different rough shapes. This one is an octahedron. The next time you look at your diamond, think about the amount of time, energy and resources have gone into making that one. Something for you to ponder about!!!
Content by courtesy of :
